Abstract
Fruit development and ripening involves a plethora of biochemical and molecular events. As such the regulation and kinetics of enzymatic reactions that are considered fundamental and central need to be understood in plants like tomato. This is because in the recent period significant interest has grown on this crop to unveil the facts with special emphasis on glutamate metabolism. Therefore there is a need to explore this particular biochemical network.
The main aim of the project is to build a model of glutamate pathway in tomato fruit during ripening, and then make sensitivity analysis to find which catalyzed reaction plays an important role in glutamate pathway and affects the accumulation of glutamate.
Differential equations were constructed and modeling softwares Copasi and Matlab were used to simulate the equations of the pathway. This model has focused on the stage from mature green (49 days after anathesis (DAA)) to post ripening (70DAA) in tomato fruits.
From the results, an increased concentrations of glutamate at 70 DAA and fluctuations in other metabolites was found which has coincided with the earlier findings. The reaction combinations (2) (7) (8) generated the highest [Glu] at 33.3 umol/ml.
Therefore, it may indicate that the enzymatic reactions concerned with the combinations (2) (7) (8) may appear vital during tomato fruit ripening and may have biotechnological applications.
Introduction
Tomato is one of the essential vegetable crops largely cultivated owing to its unique features like taste and flavor. In the recent years, this plant has drawn the attention of researchers due to its umami taste, which is considered as the fifth taste after sweet, salty, sour, and bitter (Kumiko Ninomiya, 2002).This has resulted in the preparation of good number of food items with Umami
flavor as the chief ingredient, The next important aspect of this plant is its utility as a model to study the fruit development and ripening. Fruit ripening is best regarded as the vital physiological process that reflects the involvement of various biochemical and molecular pathways which in turn are regulated by various factors, enzymes, macro and micro nutrients, light, heat and cold. Carrari et al. (2006) described a strong association between ripening associated transcripts and certain metabolic groups, such as tricarboxylic acid (TCA) -cycle, organic acids and sugar phosphates, highlighting the importance of the respective metabolic pathways during fruit development.
Hence, it may be the reason that the differences in the type and concentration of metabolites such as carotenoids, xanthophylls, chlorophylls, tocopherols, ascorbic acid, flavonoids, phenolic acids, glycoalkaloids, and saponins affect the different stages of fruit development such as green, breaker, turning pink, and red stages (Moco et al.2007).
However, to better understand the fruit ripening process it is central to primarily focus on the assimilation of nitrogen and ammonia. Nitrogen is absorbed from the soil where root hairs emerge and ammonium is in the zone of division near the apex; nitrogen absorption is induced by the presence of ammonium or nitrate (Bloom, 1996). Lam et al. (1996) reported that the inorganic nitrogen is assimilated into amino acids glutamine, glutamate, asparagine, and aspartate, which serve as important nitrogen carriers in plants. They have also reported that the enzymes glutamine synthetase (GS), glutamate synthase (GOGAT), glutamate dehydrogenase (GDH), aspartate aminotransferase (AspAT), and asparagine synthetase (AS) are responsible for the biosynthesis of these nitrogen-carrying amino acids.
Therefore, it may appear that glutamate metabolism is accompanied with the synthesis of other essential amino aicds. Hence, nitrogen may be considered to play key role during fruit ripening.
Previous workers studied the role of free amino acid content during the ripening of tomato fruit cultivars like platense, vollendung and cherry and found increased glutamate levels. They also found a decrease in glutamine synthetase and concomitant increase in NADH-glutamate dehydrogenase, suggesting a reciprocal pattern of induction between GS and GDH during tomato fruit ripening (Boggio et al. 2000).
This was supported by another report that described the concerted role of glutamine synthetase (GS) and glutamate synthase (GOGAT) in assimilating nitrogen in transgenic tobacco plant, thus demonstrating that the manipulation of GS activity has the potential to maintain crop photosynthetic productivity (Fuentes et a.l, 2001).Therefore, it may appear reasonable to assume that similar mechanism might be influencing the fruit ripening in tomato plant.
There is a strong association between glutamine synthetase activity and ammonium tolerance. This became evident from a study when several plant species including L.esculentum grown in the presence of different conditions of nitrate and ammonium, were found to accumulate equal amounts of available nitrogen, when they are able to maintain high levels of GS activity in the dark (either in leaves or in roots) and high root GDH activities (Cruz et al., 2006).Therefore, it may indicate an association between nitrogen/ammonium assimilation and GS activity that may prove beneficial for fruit ripening in tomato plants.
Earlier workers demonstrated that GS was found in red fruits of tomato plants supplemented with daily nutritional regimen containing 0.5 x Hoagland solution and described that ripe tomato fruits accumulate GS1 under high nutrition for assimilating the N-compounds (Scarpeci et al., 2007).It can be inferred that fruit ripening in tomato may be harnessed by inducing enhanced GS activity by external supplementation and facilitating ammonium assimilation.
It appears that the sole role of GS activity has become a significant aspect in the physiology of tomato fruit maturation and ripening. As such, exploring its association with the key metabolic events or pathways that drive fruit ripening may furnish better insights on the overall biochemistry of tomato plant. It would remains as a challenge to modern scientists until a thorough investigation is done on GS activity, substrate concentration, and energy requirements and its suppliers, role of coenzymes or cofactors.
Review of literature
Glutamate metabolism in tomato fruit ripening involves the integration of complex biochemical and molecular events. Earlier workers highlighted the role of various compounds in metabolic pathways underlying the development and maturation of tomato fruit. (Fernando Carrari & Alisdair R. Fernie, 2006). Prior importance was given to carbon metabolism where carbohydrates such as sucrose, glucose and fructose were indicated to present in tomato together with quinic and citric acid.
Enzymes that are proposed to play central role in developing tomato fruit are sucrose invertase and sucrose synthase (SuSy). During the earliest stages of fruit development there would be high requirement of SuSy and hexose phosphates, the rapid accumulation of starch and the highest levels of ADP-glucose pyrophosphorylase activity. From the metabolic profiling experiment, researchers have described that major hexoses, glucose and fructose were high in the cell wall components, aromatic aminoacids and aspartate, lysine, methionine, and cysteine, and in all the pigments other than chlorophyll. (Fernando Carrari & Alisdair R. Fernie, 2006).
In contrast, a decrease in the levels of TCA intermediates, sucrose, hexose phosphates, and most of the sugar alcohols was observed in red fruits. However, an increase of glutamate level was found indicating a hidden mechanism or pathway. Therefore, it may draw further attention of researchers to investigate the key contributing factors that lead to the rise of glutamate levels.
All these mentioned metabolic changes are reported to occur from development to the ripening processes in tomato fruits (Fernando and Fernie, 2006).
Earlier workers described that the two forms of enzyme glutamine synthase (GS), such as NADH dependent glutamate synthase (NADH-GOGAT) and ferredoxin-GOGAT, that catalyse glutamate were found to decrease during the ripening of tomatoes indicating an alteration in their metabolic function.
From a previous experiment, it was found that plants genetically modified with a gene for NADH-dependent glutamate dehydrogease (leg dh l) were able to synthesize high levels of glutamate compared to non-transgenic tomato plants. The amino acid levels in the transgenic tomatoes were also found to be higher (Hiroaki Kisaka, Takao Kida and Tetsuya Miwa, 2007).
Further, the strong aminating activity of NADH-GDH was also found to be correlated with the increased levels of glutamate in the transgenic tomato fruits. This correlation also coincided with increased levels of aspartate aminotransferase, NADH-malate dehydrogenase and decreased GS activity. From these earlier findings, it can be inferred that GDH is playing very important role in regulating the glutamate activity during tomato fruit ripening. In addition, high glutamate activity is also believed to have strong link with supplies of amido-type amino acids to the fruits as the source of nitrogen. Here, it can also be inferred that nitrogen/ammonium assimilation is also proved to be central in enhancing the glutamate levels in tomato fruit.
Hence, the concept of transgenics has furnished additional insights on the beneficial aspects of parallel connectivity between the free amino acid, glutamate and NADH-GDH. Therefore, GDH may have rate liming characteristic effect on glutamate (Hiroaki Kisaka, Takao Kida and Tetsuya Miwa, 2007).Next, to better facilitate this glutamate metabolism in tomato plants a cross talk between the cell organelles may be playing a role. If we initially consider the process in the chloroplast, NH3 assimilated here may be required for glutamine synthesis which in turn yields glutamate catalysed by GOGAT.
Differential equations were constructed keeping in view of the model. Since ammonia gets generated by nitrate assimilation or photorespiration, it may appear reasonable to consider it as precursor and flux producing component. Its assimilation was found to be vital for a key enzymatic step where glutamate is converted into glutamine. This reaction may be considered as the first in the glutamate metabolism and is given the constant k1.
It was reported that α-amino group of glutamate is involved in the assimilation and dissimilation of ammonia (Forde and Lea, 2007). Therefore, glutamate may be connected with aminoacid metabolism. This glutamate flux is also influenced largely by the supply of 2-OG from mitochondria. Since 2-OG is converted to glutamate by the action of GDH, and succinate by KGDH, it has significant role in glutamate metabolism in tomato plants. Other components produced from glutmate are GABA by GAD.
It was reported that GABA and amino acids such as arginine and proline are synthesized due to the supply of the carbon skeleton and α-amino group of glutamate (Forde and Lea, 2007).
In a reversible reaction GABA yields SSA catalyzed by GABAT and SSA ultimately gets associated with C3 cycle by SSADH when it contributes to the synthesis of succinate.
Therefore, it can be inferred that glutamate is not only an essential free aminoacid but also a substrate that is required for controlling and driving the metabolic events in tomato plants.
As such it is essential to connect this description of metabolism with the kinetic properties of enzymes. Initially, the catalytic conversion of glutamate to glutamine (Reaction 1) may depend on the available concentration of glutamine synthase. The more the enzyme concentration the more rapid would be the conversion of substrate into product in a given time. In other words, the velocity of glutamine synthase catalysed reaction would be directly proportional to the amount of that enzyme present. This step would be crucial to initiate the reaction. Further, according to Michaelis –Menten theory, the velocity of this reaction would increase till a definite stage when the substrate and enzyme concentrations increase.
Beyond that a saturation point would be reached where the enzyme molecules get saturated with the available substrate molecules leading to half-maximal velocity. From the plots obtained, it is obvious that the when the initial concentration was adjusted to 1mmol, a horizontal line across the middle portion of the plot was obtained.
It may indicate that a half –maximal velocity was attained indicating that both glutamine synthase and glutamate are in equivalent amounts enough to produce saturation. Hence glutamine synthase serves as parameter in the catalytic reaction of glutamate and glutamine. This is because it is strongly influencing the conversion of glutamate to glutamine and the subsequent dependant reactions.
Next, when glutamine is reconverted to glutamate the velocity of the reaction might depend on the catalyzing enzyme GOGAT. Here, the substrate glutamine may seem to largely favor GOGAT as reaction parameter because this enzyme appears vital to control the subsequent glutamate flux responsible for the production of aminoacid, alanine transferase which is reported to get upregulated during fruit ripening. Here, the transport of 2-OG from peroxisome and mitochondria is also facilitated to make 2-OGanother important substrate for GOGAT.
However, as the synthesis of 2-OG depends on C3 cycle, enzyme ICDH may appear central in driving the rate of necessary reactions in chloroplast. Further, certain significant reactions in peroxisome also contribute to the glutamate metabolism. This is facilitated initially by the transfer of glutamate from the chloroplast to peroxisome, where in association with glyoxalate it is converted to 2-OG catalysed by GGAT.
Thus, synthesized 2-OG gets transported to chloroplast where it joins with glutamate to yield glutamine. In peroxisome, glyoxolate, glutamate and serine combine under the presence of SGAT to give rise to glycine. This stage further proceeds with the transport of glycine to mitochondria where it is converted to NH3 and serine by the action of GDC and SHMT, respectively.
Ammonia gets transported to chloroplast to influence the synthesis of glutamine from glutamate under the action of GS and serine retransports to peroxisome and gets reassociated with glyoxolate.
Therefore, it can be inferred that the glutamate metabolism may involve a series of interchannel transport and resyntheses of essential substrates. This process may also be regarded as the crosstalk and may have significant role in fruit ripening. This mechanism may help in the production of additional constituents required for fruit ripening.
Ce´line Masclaux-Daubresse et al. (2006) investigated glutamate metabolism and amino acid translocation in the young and old leaves of tobacco and described that GS-GOGAT pathway and GDH play crucial roles in the nitrogen cycle of tobacco leaves. Ammonia may be considered as the ultimate form of inorganic nitrogen and well known for its assimilation into Glutamine amide group.
Therefore, a similar translocation mechanism may be playing role in tomato plants leading to an accelerated rate of free amino acid synthesis and enzyme activity that might influence fruit ripening. Previously, it was reported that the concentration of free amino acids and GDH are associated with the shelf life of tomato fruits.
This became evident when high glutamate content and inverse induction pattern of GS and GDH were detected in a standard variety of tomato plant compared to an altered variety that was found with longest SL and the lowest glutamate content. (Pratta et al., 2004).This was further strengthened by another report that described that plants transformed with a gene for GAD were found to show high glutamate content which was twice that of non- transgenic plants (Hiroaki Kisaka, Takao Kida, &Tetsuya Miwa, 2004).
To further understand fruit ripening in tomato plants it is essential to focus on ethylene which is considered as the fruit ripening hormone. Alexander and Grierson (2002) reported that ethylene-regulated ripening-related genes play important role in ethylene synthesis, fruit texture, and aroma volatile production thus indicating the importance of ethylene biosynthesis
in tomato plants. It was believed that transgenic plants created with altered ethylene biosynthesis would facilitate the development of plants with more robust or desirable traits and ethylene is mainly responsible for several plant processes such as germination, flower development, fruit ripening and responses to many environmental stimuli (Stearns & Glick, 2003).
Previously, much emphasis was given in dissecting the relationship between biochemical and genetic regulatory cascades controlling ripening, using tomato as a model species. The mechanism of cross talk between ethylene and auxin signals, and the role of ethylene-independent signals was described to play significant role in tomato fruit ripening (White, 2002).Therefore, it may indicate that gene regulation is another deciding factor that may influence glutamate network through signal transduction and the relevant expression of receptors that may have possible role. For example, large studies were carried out to highlight the benefits of ethylene receptor LeTR4 in tomato fruit ripening.
In tomato, ethylene receptors are encoded by a family of six genes and their expression is spatially and temporally controlled throughout development. It was reported that the low levels of receptor enhance ethylene responsiveness of a tissue in contrast to their higher expression that decreases ethylene sensitivity, thus indicating that receptors act as negative regulators (Klee, 2002). This was further strengthened by another report that described that reducing the levels of either LeETR4 or LeETR6 causes an early-ripening phenotype (Kevany et al., 2007).
Kevany, Taylor and Klee (2008) desribed in a recent report that fruit size, yield and flavor-related chemical composition remain unchanged during the inhibition of LeETR4 receptors thus indicating them as biological clocks regulating the onset of tomato fruit ripening. These reports would help in better identifying the candidate genes for dissecting and modifying tomato metabolic pathways.
Previous workers highlighted that the combined application of metabolic and transcriptional profiling, would help in molecular and genetic analysis of fruit development (Carrari & Fernie, 2006).
To consider other techniques, virus-induced gene silencing (VIGS) is considered as a powerful tool for the study of gene function in plants. Fu et al. (2005) described that genes associated with ethylene responses and fruit ripening, including LeCTR1 and LeEILs were silenced by this VIGS during fruit development and found that the silencing of the LeEIN2 gene resulted in the suppression of tomato fruit ripening.
Earlier workers highlighted that marker assisted breeding or QTL mapping technology has significant role in crop improvement. With these techniques a relationship between GS1 andnit rogen metabolism was well established indicating the role of certain regions of the genome that are important in regulating the activity of GS (Miflin and Habash,2002)
Since the role of GS was reported to be essential in tomato fruit ripening, application of mapping techniques may have helped in better understanding the nitrogen metabolism and production of transgenic tomato plants with different GS transgenes.
As ethylene is essential for fruit ripening it may induce relevant characteristic metabolic changes. Previous workers have reported that the inner pulp of tomatoes contained higher levels of glutamic acid, 5¢-adenosine monophosphate (AMP), 5¢-guanosine monophosphate, 5¢-uridine monophosphate, and 5¢-cytidine monophosphate.
The differences observed in the levels of these compounds may influence the umami taste difference between flesh and pulp of tomato fruits (Maria-Jose Oruna-Concha et al., 2007). Therefore, it may indicate that ethylene driven fruit ripening may determine the chief characteristic umami taste in tomato plants.Here it is reasonable to infer that gene modification techniques that focus on enhancing the umami taste for commercial applications might rely on genes or receptors that regulate ethylene biosynthesis.
Rob Alba et al. (2005) described that ethylene regulates biochemistry, morphology, and transcriptome activity throughout fruit development. They have identified 869 genes that are differently expressed in developing tomato pericarp, through transcriptome profiling via cDNA microarray analysis and reported that there are multiple points of ethylene regulatory control during tomato fruit development.
They have also found a homology sharing of genes with known gene products and signal transduction factors (Rob Alba et al. (2005). These findings may strengthen the connection between ethylene and the dependant metabolic events during tomato fruit development.
Next, it is also essential to focus on the GABA shunt as its metabolic role plays major role in carbon and nitrogen metabolism. GABA is synthesized from glutamate by the action of glutamate decarboxylase.It is latter transaminated to SSA.The synthesis of GABA is coupled with the conversion of 2-OG – glutamate. As such these three vital reactions link the shunt to several metabolic networks across subcellular compartments. It was reported that GABA gets accumulated in response to stressful conditions (Aaron Fait et al., 2007).
Therefore, it can be inferred that fruit ripening may be regulated by Glu-GABA conversion when tomato plants are exposed to biotic or abiotic stress.
Since, the conversion of glutamate to GABA is catalysed by GAD, previous researchers described its potential implications.
Hiroaki Kisaka, Takao Kida and Tetsuya Miwa (2006) described that tomato plants genetically modified with a gene for glutamate decarboxylase (GAD) in the anti sense orientation were found to accumulate glutamate in increased levels compared to non transgenic plants. Therefore, GAD was considered as the essential enzyme for the metabolism of aminoacids.They have also observed significant increases in levels of aspartic acid, serine and alanine, which belong to the glutamate family, in transgenic tomato fruits.
It may appear reasonable to connect this description with transgenic tomato plants that contain ledh gene for glutamate dehydrogenase. This enzyme was also reported to influence glutamate levels in tomato plants. Hence, these two enzymes appear crucial in controlling the levels of glutamate in tomato plants. However, understanding the kinetic behavior of these enzymes may be needed to assess the involvement of glutamate in tomato fruit ripening.
From the above example, it is obvious that the inhibition of GAD activity is leading to enhanced glutamate levels. It may indicate an inverse relationship between GAD and glutamate.
Glutamate accumulation may in turn possibly lead to increased rate of glutamine synthesis by GS. However, this glutamate accumulation may also depend on the transport of 2-OG from mitochondria and its conversion to glutamate in chloroplast, catalysed by GDH.
Therefore, GDH may be considered as the key enzyme responsible for the intercellular concentration of glutamate levels.
Gutamate dehydrogenase was reported to catalyse a reversible reaction, which could lead to either the synthesis of glutamate by amination or its catabolism by deamination.
Glutamate undergoes transamination reaction when it yields 2-oxoglutarate and other amino acids by the transfer of its α-amino group.This reversible reaction is catalysed by pyridoxal-5phosphate-containing enzymes termed aminotransferases, also known as transaminases.Thus, glutamate may act as substrate for aminotransferase reactions.
It was reported that these reversible reactions were influential in determining the stability of glutamate concentration in plants. The synthetic stage of 2-oxoglutarate from isocitrate, catalysed by isocitrate dehydrogenase may also be considered as the vital stage in the regulation of glutamate accumulation (Brian, Forde and Lea, 2007).
Debouba et al. (2006) described that tomato plants grown under different concentrations of Nacl (0,25,50 and 100mM) were found with diminished activity of nitrate reductase and glutamine synthetase,indicating the sensitivity of these enzymes involved in nitrogen assimilation, in response to salt stress.Previously, it was reported that salinity induced enhanced ethylene and CO (2) evolution rates thus shortening the time of fruit development by 4 to 15% (Mizrahi, 1982).
Therefore, it can be inferred that stress might strengthen GABA –Glu relationship and ethylene production in tomato plants during fruit ripening.
Hence, the rate of glutamate synthesis may be affected by nitrogen or ammonia assimilation, GS activity, GABA shunt subcellular transport of mitochondrial 2-OG and salinity. In addition, the key enzymes such as ICDH, GDH and GDC may also prove vital in the synthesis of free aminoacids in tomato plants. This may indicate the significance of enzyme catalysed reactions
that largely depend on the available substrate concentration. As most of these reactions appear to proceed in forward direction, an altered function or a defect in enzyme activity may induce tremendous changes on the over all metabolic pathways during fruit development.
It is reasonable to assume that an abnormal GDH activity may lead to abnormal glutamate levels and this in turn may lead to poor availability of species for reacting with the enzymes of subsequent metabolic pathways.Further, it was reported that polyamines such as putrescine, spermidine and spermine were found to occur in increased amount in the ripe pericarp of liberty variety of tomato fruit in relation to low levels of climacteric ethylene production and prolonged storage life (Saftner and Baldi, 1990).
This may indicate that tomato fruit ripening induces not only the production of essential free amino acids but also related polyamines.
Therefore, determining the amino acid levels in tomato plants may help in better understanding the underlying metabolic changes connected with fruit development.
Fernando Gallardo et al. (2004) described that in late ripening stages of tomato fruit the activity of ICDH and specific popypeptide were found to be increased in higher amount than in green fruit pericarp. This report has further suggested that enhanced ICDH activity is involved in glutamate accumulation in ripe tissues. Therefore it may indicate that the glutamate concentration in tomato fruits may also depend on the NADP+-ICDH apart from other sources previously mentioned. In addition, ICDH may also influence amino acid synthesis through large quantity of polypeptides.
Norbert Brugièrea et al. (2006) reported that GS plays major role in controlling the production of proline which is also regarded as a nitrogen source and key metabolite synthesized in response to water stress (in which plants). This mechanism may also occur in tomato plants where nitrogen metabolism and the activity of GS largely contribute to the glutamate synthesis during fruit ripening. Previous workers studied changes in metabolites during fruit development of cherry tomato and observed increased accumulation of GABA during the early 15 days after anthesis and increased GAD activity during the first 8 days of development (Dominique Rolin, 2000). These reports may further strengthen the role of GABA in the fruit development.
Further, the metabolic processes surrounding fruit development is associated with gene expression and signal transduction.
Athanasios Theologis et al. (1993) studied tomato plants constructed with antisense or sense RNA for 1 -aminocyclopropane-1-carboxylate (ACC) synthase and suggested that two signal transduction pathways function during fruit ripening.
The independent (developmental) pathway is vital for the transcriptional activation of genes such as PG, ACC oxidase, E8, E17, D2, and J49. The ethylene-dependent pathway is vital for the transcriptional and posttranscriptional regulation of genes involved in lycopene, aroma biosynthesis, and the translatability of developmentally regulated genes such as PG
(Athanasi Theologis et al., 1993). Michael Hodges (2002) reported that ammonium is made available to the plants by the GS/GOGAT pathway that requires carbon skeletons in the form of 2-oxoglutarate which is considered as a potential candidate for inducing metabolic signals to control the association of C and N metabolism. Earlier researchers have cloned a full length cDNA (legdh1) that encodes glutamate dehydrogenase (GDH) from tomato (Lycopersicon esculentum L.) and hypothesized that two GDH subunits might arise from post-transcriptional modifications of a single gene (Purnell, Stewart & Botella, 1997).
It was also obvious from the previous experiments where genes coding for glutamate decarboxylase were induced along with ripening-regulated heat-shock proteins when tomato fruits were subjected CO2 stress. This study has indicated that CO2 stress inhibits fruit ripening by preventing the expression of ripening-associated genes (Rothan et al., 1997).
This report may indicate that CO2 plays significant role in inducing a variety of genetic and metabolic changes that may affect fruit ripening. It can be also inferred that catalytic conversion of glutamate to GABA by glutamate dearboxylase or GABA shunt can be best studied by exposing plants to high levels of CO2.
Hence it is reasonable to consider CO2 as the essential and additional parameter that may decide fruit ripening. Further, to carry out all the physiological processes plants might largely depend on the selective transport of nutrients, signaling ions, and metabolites that may be facilitated through ion channels. It was described that glutamate receptors identified in Arabidopsis genome may possibly function as non-selective cation channels (NSCCs) (Romola Davenport et al., 2002).
So, it can be assumed that in tomato plants where glutamate metabolism varies with the concentration of various metabolites may posses glutamate receptors. However, further studies may be required to understand the role of plant glutamate receptors as the available information is scanty. The role of GDH was better studied by earlier researchers who employed cytoimmunochemistry and quantitative genetic approach and described that GDH protein is situated in the mitochondria of the phloem companion cells and in the cytosol of senescing organs or tissues. (Frédéric Duboisa et al., 2003).
They have also suggested that NAD (H)-GDH plays significant role in the control of plant growth and productivity. Lea and Miflin (2003) reported that GDH plays vital role as a catabolic shunt to ascertain N metabolism and facilitate the synthesis of N-rich transport compounds during nitrogen remobilization. Therefore, GDH may influence fruit development in tomato plants that is largely dependant on Nitrogen assimilation.
This study was further strengthened by another report that described the conversion of putrescine into GABA and spermidine into putrescine, indicating the presence of polyamine oxidizing enzymes in tomato pericarp tissues (Rastogi and Davies, 1990).These studies may also indicate that GABA is associated with fruit ripening through polyamine metabolism. Therefore glutamate pathway may have possible link with the levels putrescine and spermidine.
Hence, it is reasonable to consider polyamine oxidizing enzymes as additional and determining parameters of tomato fruit ripening. However, in order to better understand the importance of glutamate pathway in tomato fruit ripening there is a need to express the reactions mathematically. Previously, the process of photorespiration in plants was modeled using stochiometry of C3 plant and mathematical rate equations. For this purpose E-Cell was used as a modeling and simulation environment for biochemical and genetic processes (Pawan Dhar, Emily Wang & Yoichi Nakayama, 2001). Similar strategy if applied for glutamate network may help in thorough understanding of various enzyme catalyzed reactions, substrate concentrations and other reaction parameters.
Since carbon and nitrogen assimilation is important in all plants, earlier workers have better modeled the C/N interaction making it another important tool to explore the metabolic connections (Foyer and Verrier, 1999).Therefore, the following description is based on the Foyer and Verrier model using Copasi software to generate the out put of glutamate network in tomato fruit. This model was designed by keeping in view of principles of Michaelis –Menten and Law of mass action. Therefore, k1 values for the rest of the species were obtained in a similar manner.
It was reported that certain chemicals have ripening inducing properties. L-Methionine may have a coupling action on oxidative phosphorylation with ripening enhancing activity.DNP and CPA may also lengthen ripening by increasing the activity of polygalacturonase.It was indicated that loose’ coupling may be the mechanism by which an energy source is provided for the endergonic cell processes taking place during ripening.
Therefore, it may indicate that the mentioned agents may contribute to glutamate metabolism by exerting their fruit ripening actions. Since the aminoacid L-methionine is involved, it may also indicate that the synthesis of essential aminoacids through transamination reactions might be occurring at an increased rate in tomato plants.
Bortolotti et al (2003) reported that the key enzymes involved in glutamate metabolism are expressed differently during the ripening process. This is because, it was revealed that ripe green flesh fruits tomato contain increased glutamate levels compared to unripe or green varieties. This has also coincided with the presence and absence of GDH, respectively, in the mentioned varieties. Therefore, this could strengthen the key role of enzyme GDH during tomato fruit ripening.
The Model
Glutamate Pathway
In order to better understand the glutamate metabolism during tomato fruit ripening, a sequence of the metabolic changes needs to be considered in the form of equations. This would commence with the role of ammonia. Since it gets generated by nitrate assimilation or photorespiration, it may appear reasonable to consider it as the precursor and flux producing component. Its assimilation was found to be vital for a key enzymatic step where glutamate is converted into glutamine.
It is essential to connect this description of metabolism with the kinetic properties of enzymes. Initially, the catalytic conversion of glutamate to glutamine (k1) may depend on the available concentration of glutamine synthase.The more the enzyme concentration the more rapid would be the conversion of substrate into product in a given time. In other words, the velocity of glutamine synthase catalysed reaction would be directly proportional to the amount of that enzyme present.
It is Glu + NH3 ← k1/k2→Gln (1)
The main function of Glutamine synthetase is to catalyze the conversion of glutamate and 2-oxoglutarate to glutamine, the acidic amino acid. Glutamine synthase also known as glutamine 2-oxoglutarate amidotransferase (GOGAT) converts Glutamine to Glutamate.
This reaction involves a reductant-driven transfer of the amide group of glutamine to 2-oxoglutarate yielding two molecules of glutamate.Therefore, it can be inferred that glutamate is not only an essential free aminoacid but also a substrate that is required for controlling and driving the metabolic events in tomato plants.Next, when glutamine is reconverted to glutamate in the second reaction (k2) the velocity of the reaction might depend on the catalyzing enzyme NADPH dependant glutamate synthase (GOGAT).
Here, the substrate glutamine may seem to largely favor GOGAT as reaction parameter because this enzyme appears vital to control the subsequent glutamate flux responsible for the production of aminoacid, alanine transferase which may get upregulated during fruit ripening. Here, the transport of 2-OG from peroxisome and mitochondria is also facilitated to make 2-OG another important substrate for GOGAT.
Then the reaction is Gln + 2 OG → 2*Glu (2)
It was reported that α-amino group of glutamate is involved in the assimilation and dissimilation of ammonia (Forde and Lea, 2007).
Glutamine synthetase was believed to present in two different forms in plants. One form utilizes reduced ferredoxin (Fd) as the electron donor and the other one utilizes NADH as the electron donor. Both these enzyme forms were found to be expressed differently in separate plant tissues. Glutamine is a nitrogen containing substrates that is synthesized by an ATP consuming reaction that involves glutamate and ammonia catalyzed by glutamine synthetase. Ammonia may be generated by nitrate assimilation as observed previously or from photorespiration. Hence, it indicates that the two enzymes, glutamine synthetase and glutamine synthase are essential in metabolism of glutamate and found to catalyse irreversible reactions.
Further, certain significant reactions in peroxisome also contribute to the glutamate metabolism.
This is facilitated initially by the transfer of glutamate from the chloroplast to peroxisome, where in association with glyoxalate it is converted to 2-OG catalysed by GGAT.
Glu + Glyox ← k4/k5→ 2 -OG + Gly (3)
Thus, synthesized 2-OG gets transported to chloroplast where it joins with glutamate to yield
glutamine. 2 -OG + NH3 ← k6/k7→ Glu. (4)
Other components produced from glutamate are GABA by glutmate decarboxylase (GAD).
Glu –k8→ GABA (5)
It was reported that GABA and amino acids such as arginine and proline are synthesized due to the supply of the carbon skeleton and -amino group of glutamate (Forde and Lea, 2007).
Previously, the various significant roles of GABA as reported are that it might act as a temporary nitrogen source involved in its transport and in the regulation of cytoplasmic pH, and resistance against disease. GABA is present in tomato fruit until the late breaker stage (pink), at higher levels than glutamic acid (Pedro, 1995).
From the previous patenting information, it was understood that there are three metabolic pathways that give rise to GABA. The first pathway was associated with the catabolism of polyamines, the second with GABA aminotransferase reaction, and the most essential third pathway is through the decarboxylation of glutamate. This patent claims that plants genetically modified with a polynucleotide encoding a GAD enzyme may have their increased amounts of GABA that is essential for improved plant growth, survival and resistance to environmental stresses. However, its role in fruit ripening of tomato needs to be explored.
In a reversible reaction GABA yields succinic aldehyde (SSA) and Glutamate catalyzed by GABAT and SSA ultimately gets associated with C3 cycle by SSADH when it contributes to the synthesis of succinate.
GABA + 2-OG ← k9/k10→ SSA +Glu (6)
SSA joins C3 cycle when it is converted to succinate by succinic semialdehyde dehydrogenase (SSADH).
SSA —k10→ Sucinate (7)
After entering the C3 cycle succinate yields isocitrate.
Sucinate —k10→ Isocitrate (8)
Isocitrate —k11→ 2-OG (9)
2-OG thus formed from isocitrate further gets directly converted to sucinate.
2-OG —k12→ Sucinate (10).
Further cell wall (CW) is reported to yield isocitrate which can be considered as a constant reaction.
CW —k13→ Isocitrate = Constant (11)
Isocitrate formation from cell wall may be considered as a constant role due its role of influencing 2-OG in the subsequent reactions.
Model construction
In view of the above information a model was constructed using ordinary differential equations
(ODEs) depending on the law of mass action. The equations obtained would be explained
d [NH3]/dt = -k1[Glu][NH3] +k2[Gln] –k6[2-OG][NH3]+k7[Glu] (12)
From the above equation, it is obvious that, an increase in the concentration of NH3 in a given interval of time dt has may speed up the rate of reaction involving glutamate and ammonia, even when glutamate is present in low concentration. This would help in synthesizing glutamine at a faster rate. Simultaneously, this would also increase the rate of reaction involving 2-OG and glutamate in the presence of ammonia to yield glutamine, even when 2-OG is present in low concentration.
d [Gln]/dt = k1 [Glu][NH3] -k2[Gln] –k3[Gln][2-OG] (13)
Next, from (eq. 13) the increased concentration of glutamine obtained from the previous reaction may speed up the reversible reaction involving glutamate and ammonia to increase the
concentration of glutamine present in low amount and to facilitate the product of Gln and 2-OG that would yield glutamate.
d [Glu]/dt = k1 [Glu] [NH3] + k2[Gln] + k3[Gln] [2-OG] + k5[2-OG] [Gly] (14)
-k4 [Glu] [Glyox] + k6 [2-OG][NH3] –k7[Glu]-k8[Glu]
-k10 [Glu] [SSA] + k9 [2-OG] [GABA].
Glutamate that was obtained from the product of Gln and 2-OG (eq 13) is essential for driving the reactions involving the product of glutamate and ammonia, glutamine and 2-OG, 2-OG and glycine which are present in increased concentration.
The Glutamate concentration of chloroplast might get transported and reaches peroxisome to increase the concentration of glutamate present in low amount and combines with glyoxalate to give 2-OG which in combination with ammonia, in turn yields glutamate in low concentration.
The low concentration of glutamate may be due to the effect of retardation of 2-OG transport rate from peroxisome to chloroplast.
This Glutamate in low concentration combines with SSA. The independent concentration of 2-OG may be in increased concentration in chloroplast that ensures its combination with GABA.
Hence the concentration of glutamate plays vital role.
d [2-OG]/dt = -k3 [Gln] [2-OG] – k5 [2-OG][Gly] + k4[Glu] [Glyox] + (15)
-k6 [2-OG] [NH3] + k7 [Glu] +k10 [Glu] [SSA]
-k9 [2-OG] [GABA] +k13 [Isocitrate] – k14 [2-OG]
From the previous equations, it appears that 2-OG is an important substrate. Therefore, its increased concentration (in eq.15) helps in accelerating the low concentration of glutamine in combining with itself in mitochondria, and in ensuring the combination of the 2-OG present in low concentration in peroxisome with glycine and that of combination of glutamate and glyoxalate.
The combination of glutamate and glyoxolate in increased concentration in peroxisome might yield 2OG which subsequently transfers to mitochondria to enhance the rate of reaction involving 2- OG that may be present in low concentration to react with ammonia. This in turn may influence glutamate synthesis from glutamine (Eqs.12 & 13) and ensures the product of glutamate and SSA concentrations.2-OG present in low concentration. Glutamate in a subsequent reaction may increase the concentration of GABA which in combination with low concentration of 2-OG.
This in turn enhances the reactions leading to increased synthesis of isocitrate and low concentration of 2-OG in Citrate cycle.
d [Gly]/dt = k4 [Glu] [Glyox] –k5[2-OG][Gly] (16)
Glycine synthesized in increased amount from the previous combination of glutamate and glyoxalate (eq.15) in turn increases the rate of this combination after a cascade of previous metabolic events. Here ammonia and glutamate play vital role for ensuring the synthesis of glycine and 2 –OG in low concentration.
d [Glyox]/dt = -k4 [Glu] [Glyox] + k5 [2-OG] [Gly] (17)
The increased concentration of glyoxolate present in peroxisome increases the rate of reaction of glutamate present in low concentration and glyoxalate, and that of 2-OG and glycinecom bination.
d [GABA]/dt = k8 [Glu] + k10 [Glu] [SSA] -k9 [2-OG] [GABA] (18)
From eq. 18, GABA present in the interspaces transfers into mitochondria and induces the rate of synthesis of glutamate and succinic semialdehyde leading to their increased concentration.
It also accelerates the 2-OG concentration present in low amount and facilitates 2-OG combination with itself.
d [SSA]/dt = k9 [2-OG] [GABA] – k10 [Glu] [SSA] –K11 [SSA] (19)
The increased concentration of SSA formed from the reactants 2-OG and GABA enhances the rate of synthesis of reactant glutamate present in low concentration with SSA after a sequence of events in the citrate cycle.
d [Sucinate]/dt = k11 [SSA] – k12 [Succinate] +k14 [2-OG] (20)
Succinate formed from SSA in increased concentration facilitates the synthesis of 2-OG in high concentration in the citrate cycle.
d [Isocitrate]/dt = k12 [Succinate] – k13 [Isocitrate] + k14 [2-OG] (20)
Finally, isocitrate of citrate cycle formed from the increased concentration of succinate enhances the rate of synthesis of 2-OG.Therefore from the above differential equations it is apparent that rate of synthesis of substrates and their concentration levels seem to rely on the preceding and subsequent reactions.
The simulator software Copasi was used to model the equations.
Calculation of parameters
(The Data of the calculation are from the Carrari paper [1]).
Calculation of parameter in reaction CW⇒ Isocitrate=Constant.
The loss of CW components [CW] is obtained by calculating the change of hexose level and
[CW] =0.004 umol/ml. (showed in Table 1 in appendix file).
Calculation of reaction rate constant (k) values
- According to Table 2(showed in appendix file), we got concentrations of 7 metabolites at 4 time points from 49DAA to 70 DAA.
- According to the assumption that concentration of ammonia was not affected during this process, we got d[NH3]/dt=0.
- d[M]/dt at each time points equals the differences between each pair of time points.
For example,
d [Isocitrate]/dt at 49 DAA = Concentration of Isocitrate at 56 DAA-Concentration of Isocitrate at 49 DAA/56 days -49 days (22)
According to Table 2,
d [Isocitrate]/dt at 49 DAA = (0.24umol/ml ─ 0.18umol/ml = 0.0004 umol/ml/h)/7x 24 hours (23)
Following this approach, we can get equations of d [Isocitrate]/ dt at each time points.
at each time points.
According the Equation (21) in appendix file, we know
d [Isocitrate]/ dt at 49 DAA = k12 [Succinate] at 49 DAA – k13 [Isocitrate] at 49DAA + [CW] (24)
Then, we get
0.0004= k12*0.66 – k13*0.18 + 0.004 (25)
Using these approaches we got a set of equations of k12 and k13. Solving these equations, we can get k12 and k13 values.
Using the same way to get other equations of other k and carrying on calculations in Matlab, finally we got k values (Table 3 in appendix file) and the concentrations of SSA and Glyoxylate at 4 time points (Table 4 in appendix file). Then we got the starting concentration of metabolites (Table 5 in appendix file).
Analyses
The fruit ripening process of glutamate metabolism was further studied by making response and sensitivity analysis. Response analysis was carried out to compare the model with the actual metabolite figures from the Carrari paper.
Sensitivity analysis was carried out to observe how the glutamate levels change with the change of ammonia levels, individual changes of k values of 11 reactions when k=0 and k=100x(100 times), and the combination of sensitive reactions when k=0 and k=100x.
Results
The Glutamate metabolism in tomato fruit ripening was modeled using simulation software Copasi to carry out time course simulation, response analysis and sensitivity analysis. The results obtained were as follows.
From the data of Table 6, an increased concentration of glutamate at 70 DAA and fluctuations in other metabolites was found coinciding with the description of Carrari et al. It may indicate that the concentration of glutamate increases when the ripening process ages, thus making it an important metabolite that might influence the rate of rest of the metabolic reaction events.
According to Figure 3, the changes in plots were in agreement with the fruit ripening process and indicate their increased need. This was mostly apparent with the inclusion of three points in the (standard error) SE areas such as Glutamate, Glycine, GABA and Isocitrate. The lines of Glutamine and Succinate are inside the SE areas. In the model, the levels of 2-oxoglutarate tend to increase after 56 DAA but show fluctuations from the Carrari paper. Certain metabolite levels in also exhibit fluctuations in Carrari paper while lines are smoother in model.
In the figure 4, the lines of glutamate were in correlation with the changes in ammonia concentration that has rate limiting characteristic features. This because of change of glutamate concentration with sensitivity analysis of ammonia concentration when [NH3] =1umol/l,
10umol/ml and 100umol/ml.Ammonia concentration would appear important due to the finding that [Glu] is affected by [NH3]. From the sensitivity analysis it is also obvious that glutamate levels also changed with the individual change of k values (when k=0 and k=100x) (Figure 5 and Table 7).
The changes of glutamate levels [Glu] were further observed with the individual changes of k values (when k=0, k=100x) in the sensitivity analysis. The Glutamate concentration at 70DAA with the original k values is 9.39 umol/ml (Table 6).
According to Figure 5 and Table 7, the reactions (1),(5), (6) and (10) are sensitive for the increased glutamate levels ([Glu]) when k=0, and similarly the reactions (2),(7) (8) are sensitive when k=100x. The most sensitive is reaction (1) when k=0 and produces [Glu] at 13.2 umol/l and when k=100x it is reaction (7) that produces [Glu] at 25.5 umol/ml (Table 7). With the reaction (4) and when k=0, [Glu] level increases at the first week of ripening (49DAA-
56DAA), however it reaches the steady state early and reaches almost the same level as the [Glu] of the model. [Glu] is not affected by change of k4&k4 in reaction (3) and k15 in (11).
In order to obtain more apparent increase in glutamate level, sensitivity analysis was carried out with the combination of sensitive reactions in individual changes of k values (showed in Figure 6 and Table 8). Firstly, when k=0 sensitive reactions were combined (blue lines in Figure 6).
If enzyme Glutamate decarboxylase (GAD) was knocked out, the glutamate pathway
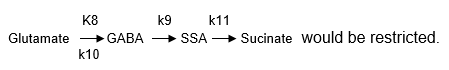
Therefore, k8=0 in reaction (5) would mean k9 & k10=0 in reaction (6), and k11=0 in reaction (7) simultaneously. Then we would get the combination (5) (6) (7). However, the concentration of [Glu] goes down obviously with (5) (6) (7). Knocking out enzymes GS, we would get combinations (1) (5) (6) (7), but it does not ensure an increase in the [Glu] levels. Then there is a need to make k14=0, and as a result enzyme KGdh would loose activity. Similarly, if both k14=0 and k11=0 which means the input of succinate was blocked, then k12=0 should occur.
Next, the combination (1) (5) (6) (7) (10) (8) with k=0 would be obtained, which could still decrease the concentrations of [Glu]. Keeping the (5) (6) (7) pathway, [Glu] grows to 14.5 umol/ml at 70DAA by just knocking out GS and KGdh with combination (1) (10).
Secondly sensitive reactions (2) (7) and (8) were combined when k=100x (red lines in Figure 6), which would mean to enhance the activity of enzymes GOGAT, SSADH and of enzymes between succinate and isocitrate, by 100 folds in Citric acid cycle.
There are four combinations: (2)(7) , (2)(8) ,(7)(8) ,(2)(7)(8) with glutamate levels at 31.6,10.2,30.9 and 33.3 umol/ml at 70DAA respectively. Finally, the combinations are from sensitive reactions when both k=0 and k=100x (black lines in Figure 6). With the knocking out of GS and over-expression of GOGAT, (1) (2) only increased [Glu] to 13.2 umol/ml while (1) (7) made [Glu] to 28.5 umol/ml. Next, [Glu] climbed to 32.1 and 32.2 by (1) (7)(8) and (10)(7)(8).
Among (1) (10) (2) (7) (8), (1)(10) and (2)(7)(8) are the most sensitive combinations when k=0 and k=100x above, and elevate [Glu] to 32.9. In conclusion, combination (2) (7) (8) generated the highest [Glu] at 33.3 umol/ml.
Table 9, has depicted the affect of two transgenic changes, KO and OE on glutamate at various stages of time period and its comparison with the model. Not all transgenic changes are found matching to the model.
Discussion
Fruit ripening process in tomato is characterized by a cascade of metabolic events. It is essential that these events comprising of reactions be modeled using sophisticated software like Copasi and Matlab. TCA intermediates mostly glutamate and other essential metabolites may be synthesized at an accelerated rate during fruit ripening. But, we need to accept that only one or two metabolites will meet the requirement and determine the rate of reactions.
As such, transgenic experiments were carried out in this regard and the results were verified. It was found that not all the reaction results of the model matched to the published observations (Table 9). This may be due to the influence of different species, time, conditions, and different varieties of tomato used. In this way many reactions of the model were evaluated and tested with the earlier findings.
For the reaction (5), the published observation matches the model, however [Glu] increases about twice in the paper of Kisaka, but 12.6/9.39 gets doubled (Table 7) for the model when GAD was knocked out. The experimental time and the varieties used may be different. It is “6 weeks after flowering” and “Lycopersicon esculentum L. “in paper of Kisaka, but “7 to 10 weeks after flowering” and “Solanum lycopersicum” for the model. Due to these two viewpoints of varieties and time, it is also acceptable that the experimental result of paper of Kisada, did not match with the model.
In reaction (4), [Glu] levels decreased slightly at 70DAA (Table 7 and Figure 5(d)) with the over expression of GDH. According to Figure 1, the reactions (1) and (5) might speed up at the same time following the increase of [Glu] caused by over expression of GDH, and the [Glu] would decrease immediately. Therefore, [Glu] did not increase finally.
This may indicate that the overexpression of GDH is differently exerting its influence on different reactions.
Further, with regard to paper of Lancien, the observation did not match due to the influence of different species. In reaction (1) of the model, [Glu] levels increased when GS was knocked out but went down slightly when GS was over suppressed (Figure 5(a) and Table 7). It is reasonable to expect that when GS was knocked out, (i.e. reaction (1) was prevented) or over expressed, the irreversible reaction (2) that involves the conversion of 2-OG and Gln into Glu might still functions.
According to Figure 7, change of [2-OG] was ignored as it was still very low. Obviously, [Glu] increased while [Gln] decreased when k=0(red lines), and [Glu] went down while [Gln] went; these changes have occurred slightly (blue lines). It is possible that when GS was knocked out, Gln would convert to Glu in reaction (2), which would enhance the [Glu]; when GS was over-expressed, due to reversible nature of reaction (1). But this did not make the result obviously.As for the reaction (3), [Glu] increased when GGAT was knocked out in paper of Igarashi (Table 9).
However, it is un-sensitive when GGAT was knocked out or over expressed (Figure 5 (c).
The reaction Glu+Glyox = 2-OG+Gly and those around Glycine and Glyoxalate which are not included in the model are reversible (Figure 8). Therefore it is reasonable to deduce that regardless of the k value changes that these reversible reactions would buildup a balanceable system.
In papers of Lancien and Joe, the results of model reflected the observations. However, due to the difference, we have to say that the match or un-match is reasonable.
In the model, according to Figure 5 (j) k14, when k14=0 in reaction (10), 2-OG Succinate was restricted and 2-OG will not lose to succinate, which may increase the 2-OG level, then increase the glutamate level. The increase in ratio is 10.7/9.4 (Table 7). The increase in ratio is 30/28 according to Table 2 of Joe’s paper.10.7/9.4 is a little bit larger than 30/28.
With regard to reaction (2), according to Figure 5 (b) k3 and Table 7, [Glu] went down when GOGAT was knocked out (which matched with the paper of Lancien and went up slightly when GOGAT was over-expressed. It is likely that as reaction (2) is irreversible, it is apparent that [Glu] will grow when GOGAT was knocking out and reduce when the enzyme gets over-expressed. So, the reason why the reduction was not obvious is that [Glu] was also affected by other reactions in this pathway of the model.
According to Figure 6, we know that combination (2) (7) (8) generated the highest glutamate levels [Glu]. [Glu] went up when GOGAT over expressing in reaction (2) (Table 7) and we have already discussed the reason in 4.1 section above.
As for the reaction (7) and (8), [Glu] went up obviously when k11 and k12=100x (Figure 5 (g) k11 and Figure 5 (h) k12 and Table 7). A simulation was carried out with this model multiple times (126DAA) to see how metabolites levels change (Figure 9).
The level of Glutamate reached steady state when [Glu] equalled about 13.7umol/ml at 126DAA (black line). [Gln] did not change obviously (red line), and [2-OG], [succinate] and [Isocitrate] were still low. However, there are obvious reductions for [SSA] (green line) and [GABA] (blue line), and they reached steady state before 126DAA. SSA and GABA were converted to Glu finally. It is likely the fact that SSADH was over expressed made [SSA] reach steady state in advance, then speeding up the reaction (7) immediately, which also made the reaction (6) faster.
Finally, the increases in the input of succinate and the reaction velocity of Citric acid cycle would increase the levels of 2-OG and then the glutamate.
It is similarly for the reaction (8). When k12=100x which would mean that the enzymes between succinate and isocitrate in Citric acid cycle were over-expressed, reaction (8), and Citric acid cycle speed up, which would increase the 2-OG level, and then the glutamate level.
In the earlier part of this report, I mentioned that the k values were obtained by calculating pairs of time points, however we get different k values when different pairs of time points are obtained. This fact needs to be discussed and may be used as a reason in order to look at the gene expression and/or protein degradation rates and to possibly incorporate these into the model.
In another earlier section, it is assumed that the model ignores the compartments and protein synthesis /breakdown. Model can be improved by considering the compartments and protein synthesis/breakdown if we get enough related data.
In the sensitivity analysis of ammonia concentration (Figure 4), we found that ammonia level is sensitive. Model can be improved if we get enough experimental data of ammonia concentration in ripening tomatoes.Cell-wall breakdown is a steady process throughout (Table 1 hexose oscillate), and isocitrate, 2-OG, Gln, GABA and glycine levels are apparently fluctuating.
This may be because, tomato is a climacteric fruit. That means It shows a massive burst of respiration on the initiation of ripening. Day 49 is mature green and the next point is breaker. So, one would expect large changes in gene expression between these points. Therefore, model can be improved if know more information about hexose oscillations in ripening tomatoes.
Acknowledgement
- Thank Prof A for tutorials
- Thank Prof B and Prof C for supply of data and information
References
Carrari, F., Baxter, C., Usadel, B., Urbanczyk-Wochniak, E, Zanor, M.I., Nunes- Nesi, A., Nikiforova, V., Centero, D., Ratzka, A., Pauly, M., Sweetlove, L.J., Fernie, AR. Integrated analysis of metabolite and transcript levels reveals the metabolic shifts that underlie tomato fruit development and highlight regulatory aspects of metabolic network behavior, 2006. Plant Physiol, 142, 1380-96.
Moco S, Capanoglu E, Tikunov Y, Bino RJ, Boyacioglu D, Hall RD, Vervoort J, De Vos RC.Tissue specialization at the metabolite level is perceived during the development of tomato fruit , (2007). J Exp Bot, 58(15-16), 4131-46.
Bloom, A.J. Life Support Biosph Sci, (1996). 3(1-2), 35-41.
Boggio SB, Palatnik JF, Heldt HW, Valle EM. Changes in amino acid composition and nitrogen metabolizing enzymes in ripening fruits of Lycopersicon esculentum Mill. (2000). Plant Sci, 159(1), 125-133.
Fuentes, S.I., Allen, D.J., Ortiz-Lopez, A., Hernández, G. Over-expression of cytosolic glutamine synthetase increases photosynthesis and growth at low nitrogen concentrations. (2001). J Exp Bot, 52(358), 1071-81.
Cruz, C., Bio, A.F., Domínguez-Valdivia, M.D., Aparicio-Tejo, P.M., Lamsfus, C., Martins-Loução, M.A. How does glutamine synthetase activity determine plant tolerance to ammonium? (2006). Planta, 223(5), 1068-80.
Scarpeci, T.E., Marro, M.L., Bortolotti, S., Boggio, S.B, Valle, E.M. Plant nutritional status modulates glutamine synthetase levels in ripe tomatoes (Solanum lycopersicum cv. Micro-Tom), (2007). J Plant Physiol, 164(2), 137-45.
Brian, G. Forde, Peter J. Lea. Glutamate in plants: metabolism, regulation, and signaling, (2007). Journal of Experimental Botany, 58, 2339–2358.
Pedro P. Gallego, Lee Whotton, Steve Picton, Don Grierson, Julie E. Gray (1995). A role for glutamate decarboxylase during tomato ripening: the characterisation of a eDNA encoding a putative glutamate decarboxylase with a calmodulin-binding site, 1995. Plant Molecular Biology, 27, 1143-1151.
Fernando Carrari & Alisdair R. Fernie. Metabolic regulation underlying tomato fruit development ,2006. Journal of Experimental Botany, 57, 1883–1897.
Ce´line Masclaux-Daubresse, Michele Reisdorf-Cren, Karine Pageau, Maud Lelandais, Olivier Grandjean, Joceline Kronenberger, Marie-He´lene Valadier, Magali Feraud, Tiphaine Jouglet, Akira Suzuki.Glutamine Synthetase-Glutamate Synthase Pathway and Glutamate Dehydrogenase Play Distinct Roles in the Sink-Source Nitrogen Cycle in Tobacco, (2006). Plant Physiology, 140, 444–456.
Hiroaki Kisaka, Takao Kida, Tetsuya Miwa. Transgenic Tomato plants that Overxpress a gene for NADH-dependent Glutamate Dehydrogenase (legdh1). (2007). Breeding Science 57,101-106.
Pratta, G., Zorzoli, R., Boggio, S.B., Picardi, L, A., Valle, E,M. Glutamine and glutamate levels and related metabolizing enzymes in tomato fruits with different shelf-life, (2004). Scientia Horticulturae, 100,341–347.
Alexander, L and Grierson, D. Ethylene biosynthesis and action in tomato: a model for climacteric fruit ripening, 2002). J Exp Bot, 53, 2039-55.
White PJ. Recent advances in fruit development and ripening, (2002). J Exp Bot, 53, 1995-2000.
Klee, H.J. Control of ethylene-mediated processes in tomato at the level of receptors (2002). J Exp Bot, 53, 2057-63.
Kevany, B.M., Tieman, D.M., Taylor, M.G., Cin, V.D., Klee, H.J. Ethylene receptor degradation controls the timing of ripening in tomato fruit, 2007. Plant J, 51,458-67.
Kevany, B.M., Taylor, M.G., Klee, H.J. Fruit-specific suppression of the ethylene receptor LeETR4 results in early-ripening tomato fruit, 2008 Plant Biotechnol J,6, 295-300.
Stearns, J.C and Glick, B.R. Transgenic plants with altered ethylene biosynthesis or perception,.2003. Biotechnol Adv, 21,193-210.
Carrari, F and Fernie, A.R.Metabolic regulation underlying tomato fruit development, 2006. J Exp Bot, 57, 1883-97.
Ben J. Miflin and Dimah Z. Habash The role of glutamine synthetase and glutamate dehydrogenase in nitrogen assimilation and possibilities for improvement in the nitrogen utilization of crops, 2002. Journal of Experimental Botany, 53, 979–987.
Maria-Jose Oruna-Concha, Lisa Methven, Heston Blumenthal, Christopher Young, Donald S. Mottram. Differences in Glutamic Acid and 5¢-Ribonucleotide Contents between Flesh and Pulp of Tomatoes and the Relationship with Umami Taste , 2007. J. Agric. Food Chem, 55, 5776-5780.
Rob Alba, Paxton Payton, Zhanjun Fei, Ryan McQuinn, Paul Debbie, Gregory Martin, Steven Tanksley and James Giovannonia. Transcriptome and Selected Metabolite Analyses Reveal Multiple Points of Ethylene Control during Tomato Fruit Development, 2005. The Plant Cell, 17, 2954–2965.
Aaron Fait, Hillel Fromm, Dirk Walter, Gad Galili Alisdair R. Fernie.Highway or by way: the metabolic role of the GABA shunt in plants, 2006.Highway Trends in Plant Science, 13, 14-19.
Debouba, M., Gouia, H., Suzuki, A., Ghorbel, M.H. NaCl stress effects on enzymes involved in nitrogen assimilation pathway in tomato “Lycopersicon esculentum” seedlings, 2006. Plant Physiol, 163, 1247-58.
Mizrahi, Y. Effect of Salinity on Tomato Fruit Ripening, 1982. Plant Physiol, 69, 966- 970.
Saftner, R.A and Baldi, B.G. Polyamine Levels and Tomato Fruit Development: Possible Interaction with Ethylene, 1990. Plant Physiol, 92, 547-550.
Fernando Gallardo, Susana Gálvez, Pierre Gadal, Francisco M. Cánovas. Changes in NADP+-linked isocitrate dehydrogenase during tomato fruit ripening, 2004. Planta, 196, 148-154.
Norbert Brugièrea, Frédéric Duboisb, Anis M. Limamia, Maud Lelandaisa, Yvette Rouxa, Rajbir S. Sangwanb, Bertrand Hirela. Glutamine Synthetase in the Phloem Plays a Major Role in Controlling Proline Production, 1999. Plant Cell, 11, 1995-2012.
Dominique Rolin, Pierre Baldet, Daniel Just, Christian Chevalier, Marc Biran and Philippe Raymond NMR study of low subcellular pH during the development of cherry tomato fruit, 2000. Australian Journal of Plant Physiology, 27, 61 – 69.
Athanasios Theologis, Paul W. Oeller, Lu-min Wong, William H. Rottmann, David M. Gantz.Use of a tomato mutant constructed with reverse genetics to study fruit ripening, a complex developmental process, 1993. Australian Journal of Plant Physiology, 27, 61 – 69.
Michael Hodges. Enzyme redundancy and the importance of 2-oxoglutarate in plant ammonium assimilation, 2002. Journal of Experimental Botany, 53, 905-916.
Purnell, M.P., Stewart, G.R., Botella, J.R. (1997). Cloning and characterisation of a glutamate dehydrogenase cDNA from tomato (Lycopersicon esculentum L.), 1997. Gene, 186, 249-54.
Rothan, C., Duret, S., Chevalier, C., Raymond, P. Suppression of Ripening- Associated Gene Expression in Tomato Fruits Subjected to a High CO2 Concentration. Plant Physiology, 114, 255-263.
Romola Davenport.Glutamate Receptors in Plants, 2002. Annals of Botany, 90,549-557.
Frédéric Duboisa, Thérèse Tercé-Laforgueb, Maria-Begoña Gonzalez-Moroc, b José-Maria Estavilloc, b, Rajbir Sangwana, André Gallaisd, Bertrand Hirel. Glutamate dehydrogenase in plants: is there a new story for an old enzyme? , 2003,Plant Physiology and Biochemistry, 41, 565-576
Peter J. Lea, and Ben J. Miflin. Glutamate synthase and the synthesis of glutamate in plants, 2003. Plant Physiology and Biochemistry, 41, 555-564.
Rastogi, R and Davies, P.J.Polyamine Metabolism in Ripening Tomato Fruit : I.Identification of Metabolites of Putrescine and Spermidine. 1990. Plant Physiol, 94, 1449-1455.
Pawan Dhar, Emily Wang, Yoichi Nakayama. Modeling Photorespiration Pathway through E-cell, 2001.Genome Informatics, 12, 306–307.
Bortolotti, S., Boggio, S.B., Delgado, L., Orellano, E.G., Valle, E.M. (2003).
Different induction patterns of glutamate metabolizing enzymes in ripening fruits of the tomato mutant green flesh. Physiologia Plantarum, 119, 384-391
Lam, H.M., Coschigano, K. T., Oliveira, I. C., Melo-Oliveira, R., Coruzzi, G.M. The Molecular-Genetics of Nitrogen Assimilation into Amino Acids in Higher Plants, 1996. Annual Review of Plant Physiology and Plant Molecular Biology, 47, 569-593.
Kumiko Ninomiya (2002). Food Reviews International, 18, 23–38.
Joe et al, Method for producing transformed plant having increased glutamate acid content. Method of producing transgenic plant with increased amino acids.
Kisaka et al, Antisense suppression of glutamate decarboxylase in tomato, 2006. Plant Biotechnology 23, 267-274.
Kisada et al. Transgenic Tomato Plant that overexpress a gene for NADH dependant glutamate dehydrogenase, 2007. Breeding science, 2007. 57, 101.
Lancien et al, Arabidopsis glt1-T mutant defenes a role for NADH-GOGAT,2002.The Plant Journal 29, 347-358.
Oliveira et al. Overexpression of Cytosolic Glutamine Synthetase, 2002. Plant Physiology 129, 1170.
Igarashi et al, Glutamate:Glyoxylate Aminotransferase Modulates, 2006 plant physiology 142, 901.